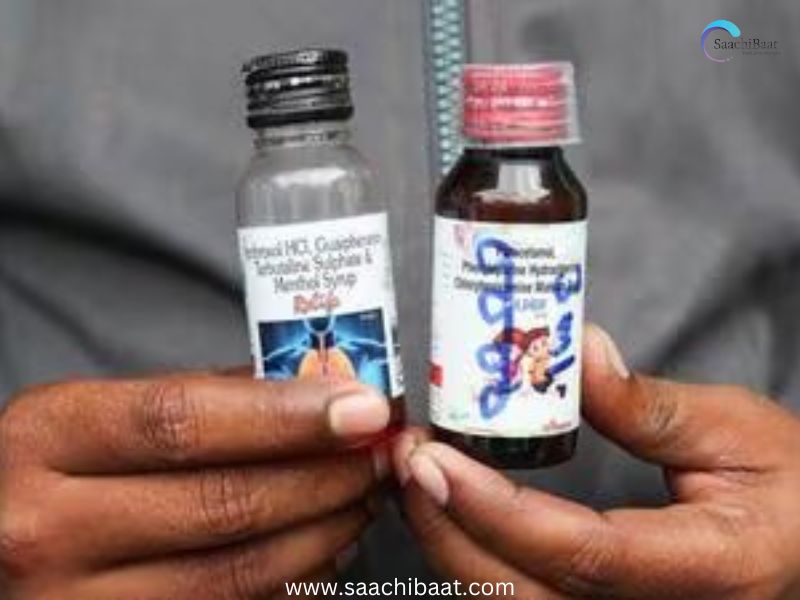
In a massive crackdown after the horrific loss of 21 children’s lives in Madhya Pradesh, the Tamil Nadu government has canceled the production license of Sresan Pharma, the firm whose cough syrup has been connected with the tragedy. The action comes after tests in labs detected toxic impurities in the drug, raising serious questions about pharma safety and regulatory control in India.
Toxic Batch Behind Multiple Deaths
The fatalities, reported in some districts of Madhya Pradesh, were originally suspected to be the result of a locally distributed cough syrup. Further investigations led back to Sresan Pharma, a Tamil Nadu-based company. The health officials confirmed that ethylene glycol and diethylene glycol, extremely toxic industrial solvents, were detected in the samples of syrup — chemicals that can lead to acute kidney failure and death, particularly among children.
A top Tamil Nadu health department official said, “The report clearly shows gross negligence and breach of safety guidelines. The company’s license to manufacture has been revoked with immediate effect pending further investigation.”
After the state action, the Union Health Ministry has ordered a nationwide inspection of the pharmaceutical units that produce pediatric syrups by all states. The Ministry has also instructed the Drugs Controller General of India (DCGI) to intensify the monitoring of small-scale drug manufacturers, who usually work with minimal quality inspections.
At the same time, the World Health Organization (WHO) has asked Indian authorities to submit a comprehensive report, citing concern about the consistent pattern of death from toxic syrup use in multiple countries. This follows similar incidents in The Gambia and Uzbekistan in 2022, both due to India-manufactured syrup contamination.
Putting India’s Drug Safety Standards in the Spotlight
The Sresan Pharma case has reopened India’s pharmaceutical regulation system to controversy, which has come under the international spotlight for quality control lapses. Experts say that though India continues to be among the largest providers of generic medicines globally, such incidents could taint India’s medical image and impact exports.
Public health activists are calling for tighter inspection, stringent punishment, and the setting up of an autonomous national drug audit organization to ensure that no such tragedy recurs.
While investigations are ongoing, the Tamil Nadu government has promised that all affected batches are being withdrawn and criminal action against the errant ones will follow. The deaths of 21 children in this tragic incident have again brought home the necessity of uncompromising pharma vigilance in the country.
××××××××××××××
Telegram Link :
For latest news, first Hand written articles & trending news join Saachibaat telegram group
https://t.me/joinchat/llGA9DGZF9xmMDc1